door bhrw | mei 14, 2026 | AGEs, wetenschap |
Promising medication which did not ‘make it’…
Part 1: Chemistry and Mechanism of Action
Advanced Glycation End Products
ALT-711, also known as alagebrium, is a pharmaceutical compound designed to address a specific biological problem: the accumulation of so-called advanced glycation end products (AGEs) in the human body. To understand how this drug works, we first need to grasp what AGEs are and why they matter.
AGEs form naturally in our bodies through a chemical process called glycation. When glucose molecules are present in our body at high levels -for instance, in people with diabetes or during normal ageing – they bond with or attach to proteins and other molecules in our tissues. These sugar-protein bonds are essentially permanent connections that stiffen and damage the structures they attach to. Some of these bonds are cross-linking bonds: the glucose bond allows two proteins to attach to each other, it acts as a sort of glue, making two protein molecules stick to each other. Over time, such AGEs accumulate in blood vessels, the heart, kidneys, and other organs, causing them to lose flexibility and function properly.
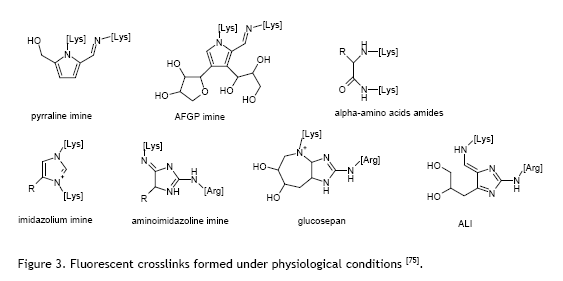
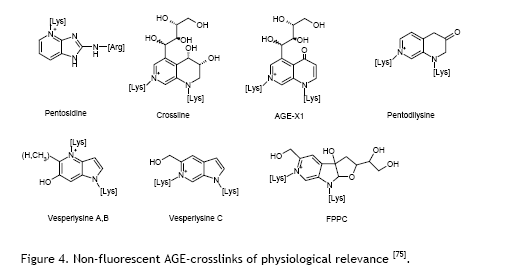
The Mechanism: Breaking Chemical Bonds
ALT-711 operates through a unique mechanism that sets it apart from other drugs. Rather than preventing new AGEs from forming (which other compounds like aminoguanidine attempt to do), ALT-711 actually breaks existing cross-links that have already formed between glucose and proteins. Think of it like untangling a knot that’s already been tied—it goes to work on damage that’s already done.
Specifically, ALT-711 cleaves carbohydrate-protein bonds that are characteristic of AGE complexes. This reversal of existing damage is groundbreaking because AGE cross-links were previously thought to be permanent. By breaking these bonds, ALT-711 can theoretically restore flexibility to stiffened tissues, particularly in blood vessels and the heart.
Why This Matters for Blood Vessels and the Heart
When AGEs accumulate in large arteries, they cause arteries to stiffen and lose their natural elasticity. Normally, arteries are flexible tubes that expand and contract with each heartbeat, helping to pump blood throughout the body. When AGEs cause stiffening, this natural pumping action becomes compromised. By breaking AGE cross-links, ALT-711 can restore large artery compliance—essentially returning flexibility and volume capacity to blood vessels. This improved elasticity means blood vessels can function more like they should, potentially reducing strain on the heart and improving overall cardiovascular function.
Chemical Classification
ALT-711 represents a first-of-its-kind class of drugs called AGE crosslink breakers. Unlike previous approaches that focused on slowing AGE formation, this compound targets the fundamental structural problem: the rigid bonds that make tissues stiff and dysfunctional.
The significance of this approach cannot be overstated. If successful, it would represent a therapeutic paradigm shift—moving from prevention to reversal in addressing ageing-related vascular stiffness and dysfunction.
References:
* Treskes M, Wolffenbuttel BHR. Lange termijn schade van diabetes mellitus: de rol van irreversibele glyceringsproducten. Ned Tijdschr Geneeskd 1996; 140: 1299-1303.
* Wolffenbuttel BHR, Boulanger CM, Crijns FRL, Huijberts MSP, Poitevin P, Swennen GNM, Vasan S, Egan JJ, Cerami A, Lévy BI. Breakers of advanced glycation endproducts restore large artery properties in experimental diabetes. Proc Natl Acad Sci USA 1998; 4630-4.
* Mentink CJ, Hendriks M, Levels AA, Wolffenbuttel BHR. Glucose-mediated cross-linking of collagen in rat tendon and skin. Clin Chim Acta 2002; 321: 69-76.
Source for the figures:
* Mentink, C. Thesis AGEs and diabetic complications. Maastricht, 2006
* Ulrich P, Cerami A. Protein glycation, diabetes, and aging. Recent Prog. Horm. Res. 2001;56:1-21.
Part 2: Animal Studies
Evidence of Promising Preclinical Results
The available literature indicates that ALT-711 showed promising results in animals before human trials began. These experimental and preclinical studies demonstrated that the drug could improve cardiac function and reduce symptoms in heart failure models. The fact that researchers proceeded from animal studies to human clinical trials suggests that the preclinical safety and efficacy data were sufficiently encouraging to warrant further investigation.
Mechanism Validation in Animal Models
Animal studies focused on validating the proposed mechanism of action—that breaking AGE cross-links would actually restore tissue flexibility and improve organ function.
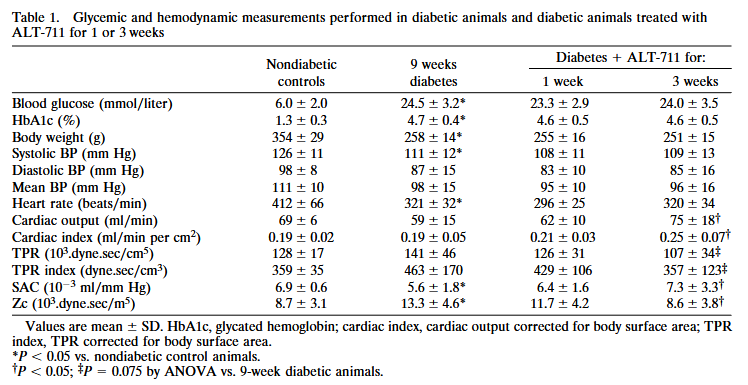
My group did an extensive study into the mechanism of action of ALT-711, in which we evaluated the effects of treatment with ALT-711 on functional arterial wall properties in diabetic rats. This work was done in the department of Professor Bernard Levy in Paris. Male Wistar rats were made diabetic at the age of 9–10 weeks by i.p. injection of 70 mg/kg of streptozotocin. Only animals that developed blood glucose levels >15 mmol/liter were used. After 9 weeks of diabetes, the animals were divided into three groups: one group was studied to assess the exact hemodynamic changes that were caused by the diabetic state (n = 13), and two groups received ALT-711 (1.0 mg/kg per day i.p.) for either 1 or 3 weeks (n = 8–10 animals in each group) to assess possible reversal of the diabetes-induced cardiovascular abnormalities by treatment with this compound.

The different approaches used in these studies consistently show the beneficial effects of treatment with the AGE breaker ALT-711 on arterial elasticity. In vivo, treatment increased systemic arterial compliance (SAC) and reduced characteristic aortic input impedance (Zc). There was a decrease in carotid artery stiffness, as assessed with ultrasound in vivo and in vitro. These effects seemed to be related to the duration of the treatment, with stronger effects after 3 weeks than after 1 week. The finding of the marked increase in compliance and decrease in impedance cannot be attributed to differences in BP, which did not change during treatment; therefore, it reflects intrinsic modifications of the mechanical properties of the arterial wall. The increase of SAC and decrease of Zc indicate that because of treatment with ALT-711 the stiffness of the aorta was reduced by 25% (SAC) to 35% (Zc) to levels comparable to those observed in nondiabetic animals. This improvement was comparable to our results achieved with preventive treatment with aminoguanidine in diabetic rats.
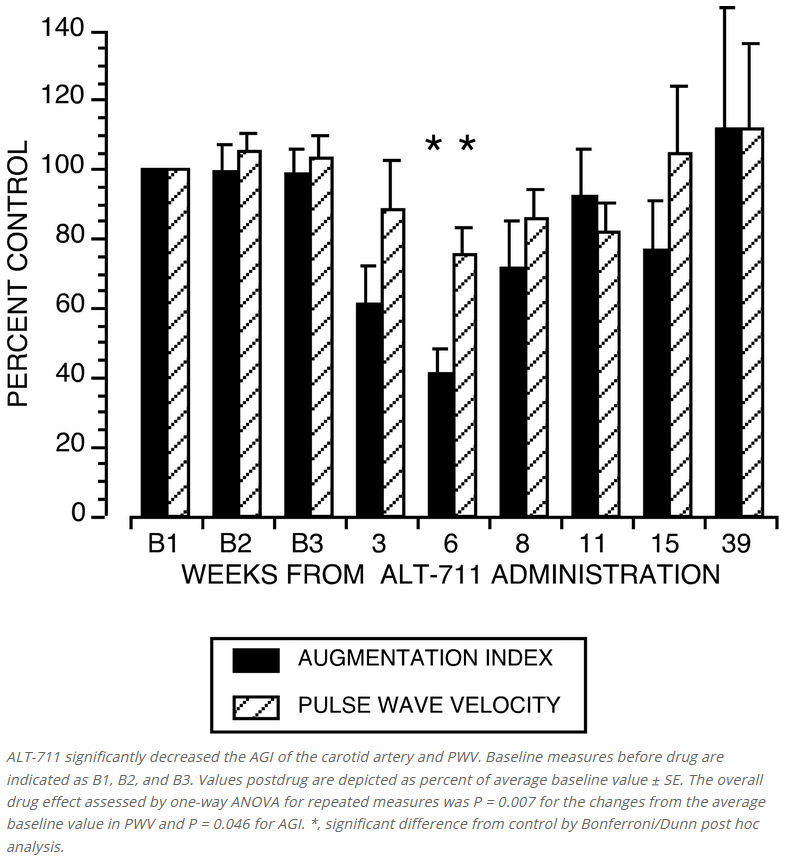
In healthy older monkeys without diabetes, ALT-711 improved both arterial and ventricular function and optimised ventriculo-vascular coupling (https://pubmed.ncbi.nlm.nih.gov/11158613/).
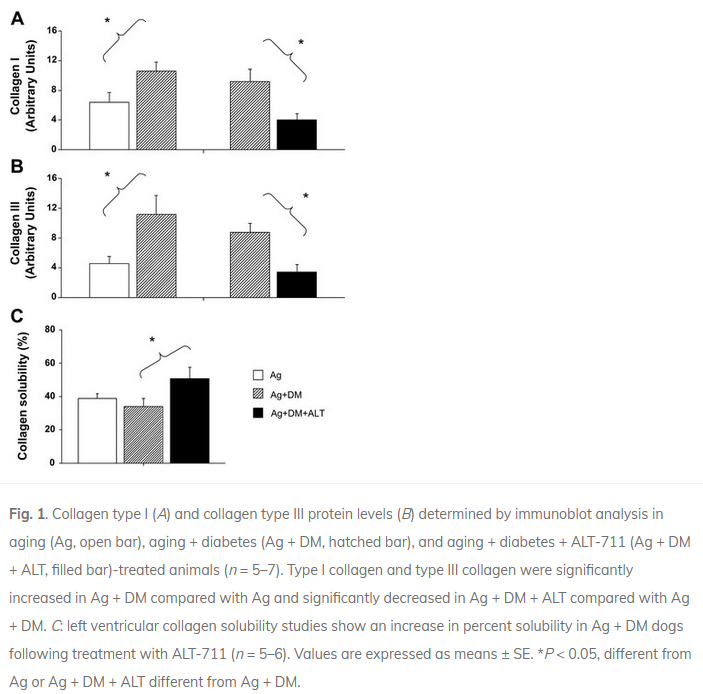
In aged dogs, 1 month of administration of ALT-711 yielded a significant reduction (approximately 40%) in age-related left ventricular (LV) stiffness. This decrease was accompanied by an improvement in cardiac function (https://pubmed.ncbi.nlm.nih.gov/10706607/). Furthermore, in Sprague-Dawley rats made diabetic with a streptozotocin injection, ALT-711 treatment restored LV collagen solubility and cardiac BNP, in association with reduced cardiac AGE levels and abrogation of the increases in RAGE, AGE-R3, CTGF, and collagen III expression (https://pubmed.ncbi.nlm.nih.gov/12623881/). And a study in dogs confirmed these beneficial effects on the heart. Diabetes induced in the aging heart decreased LV systolic function (LV ejection fraction fell by 25%), increased aortic stiffness, and increased collagen type I and type III protein content. ALT-711 restored LV ejection fraction, reduced aortic stiffness and LV mass with no reduction in blood glucose level, and reversed the upregulation of collagen type I and type III. Myocardial LV collagen solubility (%) increased significantly after treatment with ALT-711 (https://pubmed.ncbi.nlm.nih.gov/12946933/).
Part 3: Clinical Studies in Humans
will follow soon
Prepared with assistance by Perplexity.
door bhrw | apr 9, 2026 | gezondheidszorg, social media, wetenschap |
Altmetric: een moderne manier om de impact van wetenschappelijk onderzoek te meten.
Stel je voor: je bent arts of wetenschapper en je publiceert een artikel over een nieuw medicijn of een klimaatmodel. Traditioneel meet je succes aan de hand van citaties in andere wetenschappelijke artikelen – hoe vaak anderen je werk aanhalen. Maar dat duurt jaren en zegt weinig over wat er buiten de academische wereld gebeurt. Hoe weet je of journalisten erover schrijven, of het op sociale media besproken wordt, of hoe en wanneer beleidsmakers het gebruiken? Hier komt Altmetric om de hoek kijken. Dit programma, gebaseerd op altmetrics (alternatieve meetmethoden), volgt de online aandacht voor iemand’s onderzoek en geeft een breder beeld van de impact.
Altmetric is een systeem dat automatisch data verzamelt uit tientallen bronnen op internet. Denk aan sociale media zoals Twitter, BlueSky en Facebook, waar mensen je artikel delen of bespreken. Het haalt ook info van kranten als The Guardian of New Scientist, blogs van onderzoekers of organisaties zoals Cancer Research UK, en tools als Mendeley waar wetenschappers artikelen opslaan en aanbevelen. Zelfs niet-Engelstalige media en beleidsdocumenten worden meegenomen. Het slimme is dat Altmetric ‘linkt’ herkent: een tweet over een samenvatting van een wetenschappelijk artikel op PubMed, een krantenartikel via de uitgever en een blog – allemaal over hetzelfde artikel – worden samengevoegd tot één overzicht.
Het hart van Altmetric is de zogenaamde Altmetric Attention Score, een getal in een kleurrijke ‘donut’ of badge die je vaak ziet bij artikelen op websites van tijdschriften. Deze score vat de hoeveelheid en kwaliteit van de aandacht samen. Een algoritme berekent het door factoren mee te wegen zoals het bereik van de bron (een tweet van een beroemde professor telt zwaarder dan een random post), de taal, en of het positief of negatief is. Het is geen perfecte wetenschap – het is een momentopname die continu update – maar het geeft snel inzicht in hoe je werk resoneert.
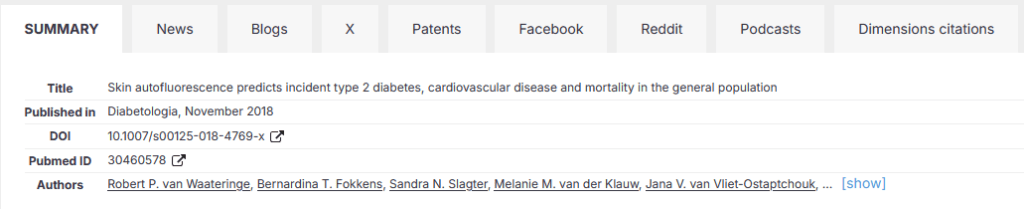
Een voorbeeld, aan de hand van één van onze artikelen:
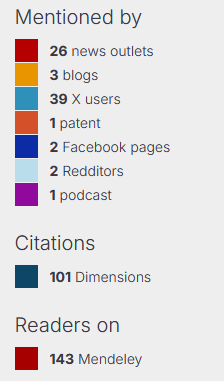
Voor wetenschappers biedt dit enorme voordelen, vooral in een tijd waarin ‘open science’ en maatschappelijke relevantie steeds belangrijker worden. Traditionele metrics zoals de Journal Impact Factor (JIF $) berekenen vooral citaties in topbladen en negeren boeken, blogs of praktijkimpact. Altmetrics corrigeren dat: ze tonen niet alleen berichten over een artikel, maar ook hoe je onderzoek de samenleving raakt – via nieuws, beleid of publiek debat. Bijvoorbeeld: scoort je artikel hoog op Twitter? Dan bereikt het veel niet-wetenschappers, mensen van patiënten organisaties, en misschien ook policymakers. Er zijn steeds meer mensen die bijvoorbeeld preprint-servers (websites waar artikelen worden verzameld / gedeponeerd die nog niet door reviewers beoordeeld en gepubliceerd zijn) volgen en screenen wanneer weer een artikel wordt gepubliceerd over hun favoriete onderwerp. Wordt het genoemd in een rapport? Dat wijst op praktische toepassing.
Met de gratis Altmetric-bookmarklet (een browser-extensie) check je direct de score van elk artikel via DOI * of PMID **. Instellingen zoals universiteiten hebben vaak toegang tot Altmetric Explorer for Institutions (EFI), een dashboard waar je zoekt op auteur, afdeling of groep. Zo zie je welke papers van jouw groep het meest besproken worden, inclusief sentimentanalyse (positief/negatief) en demografische details over wie erover praat. Handig voor grant-aanvragen: “Kijk, ons werk werd opgepikt door 50 media en 200 tweets!” Het helpt ook om trends te spotten, zoals groeiende aandacht over de tijd, en om te netwerken met wie je werk deelt.
$ JIF: De Journal Impact Factor (JIF), ook wel impactfactor genoemd, is een maat die de gemiddelde citatiefrequentie van artikelen in een wetenschappelijk tijdschrift aangeeft. Hoe hoger de JIF, hoe vaker artikelen uit dit tijdschrift geciteerd worden door andere onderzoekers.
* DOI: een Digital Object Identifier (DOI) is een unieke, permanente alfanumerieke code die wordt toegewezen aan digitale objecten, zoals wetenschappelijke artikelen, datasets en rapporten. Het functioneert als een “digitale vingerafdruk” die zorgt voor duurzame vindbaarheid en citeerbaarheid.
** PMID: een PMID (PubMed Identifier) is een uniek nummer dat door de National Library of Medicine (NLM) in de VS wordt toegewezen aan elk artikel in de PubMed-database (https://https://pubmed.ncbi.nlm.nih.gov/). Met dit unieke nummer kun je wetenschappelijke biomedische publicaties snel en accuraat terugvinden, zonder de volledige titel of auteursnaam nodig te hebben.
Maar die sentiment analyse, die blijkt vaak niet te kloppen. Althans, ik bekeek dit voor enkele negatieve “beoordelingen” door Altmetric. In meerdere gevallen werd de referentie naar één van mijn publicaties, gebruikt om een bewering van iemand te ontzenuwen, als negatief beoordeeld. Ook hier een voorbeeld: ons NHANES-artikel krijgt een negatieve beoordeling van Altmetric op basis van deze tweets. Onbegrijpelijk.:

Altmetric geeft daarom terecht een uitgebreide disclaimer:
“This feature uses AI to assign sentiment scoring to posts that mention research. Although the quality of AI outputs is often high, they may contain incorrect information. The validity of the output should be verified by reviewing the referenced documents.”
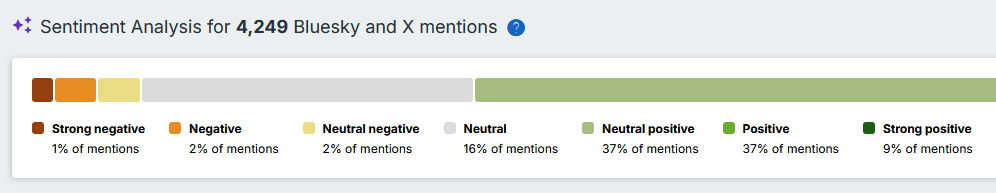
Het is mogelijk om een negatieve beoordeling van een kanttekening te voorzien, maar het is mij volstrekt onduidelijk wat Altmetric precies met deze feedback doet.
Altmetric is een aanvulling, geen vervanging voor citaties. Het is vluchtig – een hype op social media kan morgen weg zijn – en niet altijd betrouwbaar voor evaluaties. Experts waarschuwen: gebruik het kwalitatief, naast expert-oordeel, zoals in het Leiden Manifesto voor onderzoekmetrics (https://www.leidenmanifesto.org/). Focus niet blind op getallen; ze ondersteunen verhalen over bereik en invloed. Desondanks transformeert het de wetenschap: het maakt impact meetbaar op het ‘sociale web’, waar onderzoek sneller leeft en breder landt.
Samenvattend, Altmetric helpt wetenschappers hun werk zichtbaarder te maken, en geeft inzichten in zaken als impact en relevantie. In een wereld waar wetenschapsfondsen en universiteiten steeds meer waarde hechten aan maatschappelijke impact, is dit veel waard. Het moedigt artsen en onderzoekers aan om gegevens en resultaten van wetenschappelijk onderzoek te delen, breed te communiceren en te laten zien dat wetenschap niet alleen in ivoren torens blijft, maar de hand reikt naar de echte wereld.
Links:
https://libguides.ru.nl/medisch/bibliometrie/
https://mindthegraph.com/blog/nl/altmetric/
https://library.maastrichtuniversity.nl/database/altmetrics/
https://uba.uva.nl/content/nieuws/2025/01/library-research-hack-altmetric-explorer.html?cb
door bhrw | mrt 12, 2026 | B12, gezondheidszorg, wetenschap |
Some people consider vitamin B12 as a simple vitamin, which only affects the synthesis of red blood cells (figure 1, picture by Grok).
 So, if you do not have anaemia, you don’t have a B12 deficiency, and outside the setting of ‘anaemia’, B12 is useless. This is absolutely not correct. Vitamin B12 is a very important compound, it has multiple effects in our body, and is especially needed for optimal nerve function and for well-functioning mitochondria, the small organelles in our cells that are responsible for making energy.
So, if you do not have anaemia, you don’t have a B12 deficiency, and outside the setting of ‘anaemia’, B12 is useless. This is absolutely not correct. Vitamin B12 is a very important compound, it has multiple effects in our body, and is especially needed for optimal nerve function and for well-functioning mitochondria, the small organelles in our cells that are responsible for making energy.
B12 -as a medication- has also been used by intensive care doctors in severely ill people. There are several interesting papers on the use of hydroxocobalamin for people who suffer from (refractory) vasoplegia after cardiopulmonary bypass surgery, heart surgery. Vasoplegia (vascular shock) is a clinical syndrome characterised by severe vasodilatation (widening of the blood vessels) of the smaller arteries and arterioles, hypotension (very low blood pressure), and low systemic vascular resistance that does not respond to treatments with multiple vasopressor medications, medications that should promote the vessels to get narrower and blood pressure to increase. I have selected three papers in this area to explain this complex medical topic further.

The 2018 Shah paper, see figure 2:
Full reference: Shah PR, Reynolds PS, Pal N, Tang D, McCarthy H, Spiess BD. Hydroxocobalamin for the treatment of cardiac surgery-associated vasoplegia: a case series. Can J Anaesth. 2018 May;65(5):560-568. English. doi: 10.1007/s12630-017-1029-3. Epub 2017 Dec 5. PMID: 29209927.
Link to the paper: https://link.springer.com/article/10.1007/s12630-017-1029-3
In this paper, already published 8 years ago (!), doctors from Richmond, Virginia, report their experience with using a hydroxocobalamin (B12) infusion as a potential rescue adjunct for refractory vasoplegia during cardiopulmonary bypass surgery. They studied a series of 33 individuals, who underwent cardiac surgery between 1 January 2013 and 31 December 2015. Patients were given intravenous infusion of methylene blue or vitamin B12 (hydroxocobalamin) for severe not-responsive hypotension during, or immediately following, surgery.
Compounds as methylene blue and hydroxocobalamin are mechanistically unique and thought to work through nitric oxide–based mechanisms to induce vasoconstriction. When you ask Google’s AI, it reports that Vitamin B12 (cobalamin) also acts as a direct scavenger of reactive oxygen species (ROS), particularly superoxide (O2-), with activity comparable to that of superoxide dismutase (SOD). It reduces oxidative stress by scavenging (‘hunting and grabbing’) radicals and preserving glutathione, protecting DNA and cells from damage. It is effective in mitigating ischaemia/reperfusion injury and supporting neural health.
In the study by Shah et al, individuals had received methylene blue and/or hydroxocobalamin (5 g, Cyanokit), administered as an intravenous infusion over a 15 minute period. The overall finding and conclusion of the paper is that there was considerable diversity in the response of an individual to B12; the authors suggested that this may have depended on both the patient’s preoperative condition and the fact that the timing of the administration was not standardized. Nevertheless, in their opinion, vitamin B12 may provide a useful alternative or additional therapy to treat refractory hypotension and vasoplegia, but -as they were looking back- they state that prospective controlled clinical trials to assess the true efficacy of the therapy are needed.

A second paper on this topic (figure 3) was published in 2025 by a group of doctors in Cairo, Egypt, and it describes the prophylactic use of hydroxocobalamin in patients at high risk of developing vasoplegia after cardiopulmonary bypass. So, a high dose of B12 is administered to assess whether this can prevent severe illness.
Full reference: Salah D, Ahmed S, Ibrahim DA. Use of Hydroxycobolamin in the Prevention of Vasoplegic Syndrome in Adult Patients Undergoing Cardiopulmonary Bypass: A Controlled Prospective Trial. J Cardiothorac Vasc Anesth. 2025 May;39(5):1180-1187. doi: 10.1053/j.jvca.2025.02.001. Epub 2025 Feb 5. PMID: 40055025.
Link to the paper: https://www.jcvaonline.com/article/S1053-0770(25)00114-4/abstract
How did they treat the participants? At the end of cardiopulmonary bypass (CPB), the participants were assigned by chance into two groups: 30 individuals in group I received hydroxocobalamin 5 g intravenously via the central venous catheter as a bolus over 15 minutes reconstituted in 200 ml of normal saline, and 30 individuals in group II (the so-called ‘control group’) received 200 mL of normal saline intravenously over 15 minutes. When they collected all information, it turned out that the B12 prevention group showed higher arterial blood pressure 30 and 60 minutes after CPB separation, they required lower amounts of vasopressor treatment (norepinephrine) at minutes 30 and 60 after CPB separation, as well as total dose norepinephrine equivalent. Also, lower serum lactate concentrations (a sign of poor blood perfusion of the body’s tissues), and lower incidence of vasoplegic shock syndrome and norepinephrine-resistant refractory vasoplegia were noted. The number of days that a patient neededa rtifical ventilation (3.8 vs 4.1 days), the length of stay in the intensive care unit (6.2 vs 7.0 days), the length of the stay in the hospital, and the number of participants who did not survive this severe illness (1 of 30 vs 3 of 30) were lower or less frequent in the B12 group, but the authors note that these differences were not statistically significant.
The third paper which we discuss briefly, is a recent article from a group of doctors in Pittsburgh, PA, USA; figure 4 shows what we call the graphical abstract of this paper.
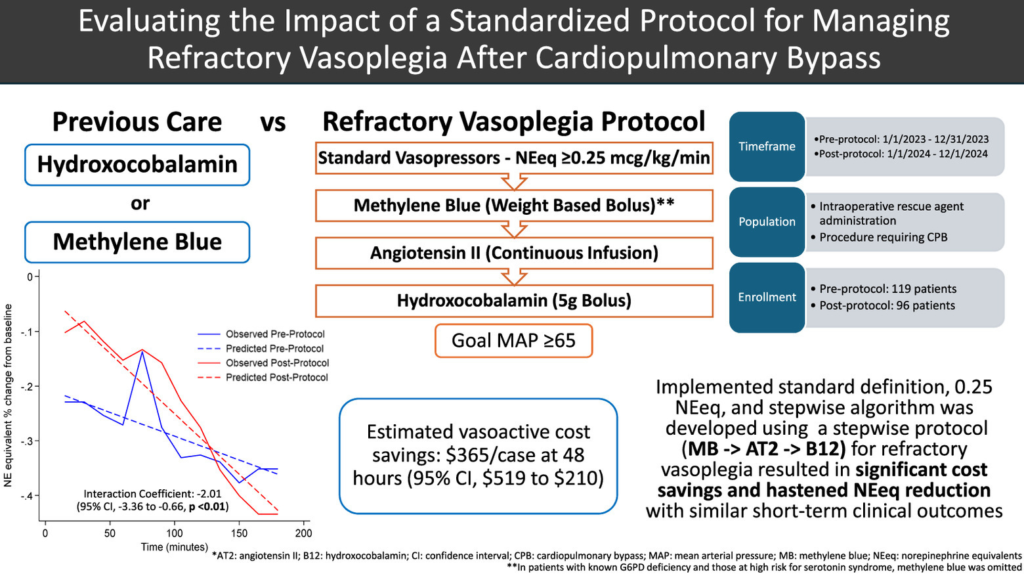
Full reference: Teletnick A, Suh K, Boisen M, Brown JA, Cabral B, Murray H, Paley C, Sullinger D, Sultan I, Thoma F, Williams J, Subramaniam K, Rivosecchi RM. Evaluating the Impact of a Standardized Protocol for Managing Refractory Vasoplegia After Cardiopulmonary Bypass. J Cardiothorac Vasc Anesth. 2026 Feb 6:S1053-0770(26)00110-2. doi: 10.1053/j.jvca.2026.02.003. Epub ahead of print. PMID: 41765732.
Link to the paper: https://www.jcvaonline.com/article/S1053-0770(26)00110-2/fulltext
This article describes a prospective evaluation of the haemodynamic impact and cost-effectiveness of a standardized protocol for treating people who develop refractory vasoplegia after cardiopulmonary bypass. In this paper, refractory vasoplegia was defined as at least 0.25 μg/kg/min norepinephrine equivalents (NEeq), and a stepwise rescue treatment protocol was developed, starting with methylene blue, then giving angiotensin II and subsequently hydroxocobalamin (see figure 4).
The authors assessed the effects of the rescue intervention by looking back in time and comparing patients undergoing CPB after the implementation of this new protocol with those undergoing CPB before protocol implementation, the latter group receiving rescue agents at the discretion of the managing ICU doctor. Again, the hydroxocobalamin dose was 5 g, the so-called Cyanokit.
Their findings in short: they observed a faster reduction in vasopressor therapy and lower associated costs of the treatment with the rescue intervention. They did, however, not find any differences in short-term clinical outcomes between groups.
So, to summarize these three papers, it appears that hydroxocobalamin has an important role in treating the so-called vasoplegia in critically ill patients after heart surgery. However, several additional questions arise. For example, is there an optimal dose of hydroxocobalamin, and, also, can repeated administrations of B12 add to a prolonged or sustained effect? The findings of the Egyptian group regarding shorter durations of mechanical ventilation and shorter stays in the intensive care unit and in the hospital are very interesting and warrant further investigation.
Another interesting aspect to consider is that of pre-operative B12 status. As a doctor who is interested in the effects of B12 deficiency, I ask myself: do individuals who are undergoing surgery while having B12 insufficiency or deficiency have a higher risk of becoming severely ill and developing vasoplegia? Especially now that we see more and more people adopting a plant-based diet, who sometimes develop B12 deficiency despite supplementation. I hope that the results described in these three papers will encourage more researchers and clinicians to further explore the intriguing effects of B12.
Parts of this blog have also been published in CluB-12’s March 2026 Newsletter.
For translation into Dutch, copy this blog to Google Translate
door bhrw | mrt 5, 2026 | B12, gezondheidszorg, wetenschap |
Amsterdam kende vroeger een eminent hoogleraar Neurologie, professor Arie Biemond (1902-1973), van wie uitstekende kennis, brede klinische ervaring, grote voorkomendheid en enthousiasme de belangrijkste kenmerken waren. Dit kunnen we teruglezen in een uitgebreide beschrijving van zijn persoon en klinische werk, geschreven door twee van zijn voormalige assistenten, dr. Frederiks en Blomjous (https://www.neurologie.nl/wp-content/uploads/2022/01/History-of-Neurology-in-the-Netherlands-hoofdstuk-17-blz-283-290.pdf). Dit blog is mede een eerbetoon aan de klinische kennis die professor Biemond had ontwikkeld over de desastreuze gevolgen van vitamine B12 tekort voor het zenuwstelsel.

Klinische lezing
Ik was nog lang niet geboren, toen professor Biemond in 1953 een uitstekende lezing gaf over de afwijkingen van het centrale zenuwstelsel bij pernicieuze anemie. De lezing is nog terug te vinden in één van de uitgaven van de serie Het Hormoon, een serie die jarenlang is uitgegeven door de N.V. Organon, en zich wijdde aan alle aspecten van de klinische endocrinologie en stofwisselingsziekten. In de samenvatting van zijn lezing geeft hij een zeer goed leesbare en gedetailleerde beschrijving van de neurologische afwijkingen bij pernicieuze anemie en vitamine B12 tekort. De beschrijvingen zijn zo precies en welomschreven, dat je je afvraagt waarom deze kennis zo in het slop is geraakt de afgelopen decennia.

Symptomen
Een paar voorbeelden uit de tekst. Allereerst gaat Biemond uitgebreid in op de symptomen. Hij geeft hierbij aan dat klachten van neuropathie vaak centraal staan in de eerste fase van schade aan het zenuwstelsel door B12 tekort. “Paraesthesieën van wisselende intensiteit aan alle vier de extremiteiten. Zij ontbreken nimmer en gaan in de regel vooraf. Zij localiseren zich niet uitsluitend aan vingers en tenen, doch aan handen, voeten en onderbenen, eventueel aan de extremiteiten in hun geheel. Ofschoon zij door de patiënten op verschillende wijzen worden omschreven, overheerst toch de sensatie van doofheid en van het gevoel, dat de huid over bepaalde gebieden door een laagje stof (of vilt of gips) is bekleed. In deze vorm hebben de paraesthesieën stellig een belangrijke diagnostische betekenis. Zij vormen het eerste teken, dat de achterstrengen van het ruggemerg niet normaal meer functioneren. “
Over het al dan niet samengaan met anemie schrijft hij: “Er kan zich een typische gecombineerde strengziekte ontwikkelen, wanneer het bloedbeeld nog nauwelijks of in het geheel geen tekenen van anaemie vertoont. De pernicieuze anaemie is dan nog slechts in latente vorm aanwezig, doch wel pleegt regelmatig achylia gastrica, die histamine-refractair is, te worden gevonden.”
Hierover vervolgt hij: “De in de kliniek zo veelvuldig voorkomende discongruentie tussen anaemie en ruggemergsdegeneratie bij éénzelfde grondoorzaak heeft geleid tot de invoering van de begrippen “haematopoietine” en “neuropoietine” ….” Hierbij “moet in het bijzonder het werk van CASTLE worden vermeld, die in 1929 de begrippen “intrinsic” en “extrinsic factor” invoerde, waarvan de eerstgenoemde in het normale maagsap in voldoende mate aanwezig is. Door de onderzoekingen der laatste jaren is thans wel komen vast te staan, dat de extrinsic factor identiek is met vitamine B12 en dat deze stof, na binding met de intrinsic factor, op bloedbeeld en centraal zenuwstelsel op gelijke wijze uitwerkt en dus zowel “haematopoetisch” als “neuropoetisch” is.”
Met andere woorden, wat hij hierbij duidelijk uitlegt, is dat vitamine B12 tekort bij de één leidt tot neurologische klachten, en bij de ander tot bloedarmoede. Hierbij moeten we aantekenen dat om meerdere redenen het klinisch beeld van pernicieuze anemie de afgelopen decennia is veranderd, zowel door vroegere diagnostiek als door veranderingen van bv. voedingsgewoonten. Tegenwoordig wordt bloedarmoede bij minder dan 20% van de mensen met pernicieuze anemie gevonden. Waarom dat precies is, weten we niet heel goed, maar er zijn diverse mechanismen die mogelijk een rol spelen. Eén ervan is de inname van foliumzuur. In de jaren 50 van de vorige eeuw waren B12 injecties nog niet breed beschikbaar, en werden nogal wat mensen met bloedarmoede bij pernicieuze anemie behandeld met foliumzuur. Dit was heel effectief in het corrigeren van de bloedarmoede, maar leidde bij veel mensen tot het ontstaan of verergeren van klachten van neuropathie. Deze behandeling is daarom weer snel verlaten toen voldoende B12 in injectievorm beschikbaar kwam. Een jaar of 20 a 25 geleden zijn een aantal landen, waaronder de Verenigde Staten, overgegaan op het toevoegen van foliumzuur aan diverse voedingsmiddelen. Foliumzuurtekort is hierdoor uitgebannen, bijna iedereen in de VS heeft een normale folaat hoeveelheid in zijn of haar lichaam. Het is goed mogelijk dat juist hierdoor, bij iemand die B12 tekort ontwikkelt, bloedarmoede veel minder vaak ontstaat, maar neuropathie en neurologische klachten juist vaker. Een andere factor is oxidatieve stress. Google beschrijft het als volgt: “Oxidatieve stress is een chronische toestand van verhoogde vrije radicalen en verminderde antioxidantverdediging, die (onder andere) nauw verbonden is met obesitas. Vetweefsel bij obesitas werkt disfunctioneel, wat leidt tot ontstekingen, zuurstoftekort en verhoogde vetzuren. Dit proces beschadigt cellen, draagt bij aan insulineresistentie en verhoogt het risico op hart- en vaatziekten en diabetes.“ En we zien in de hele wereld nog altijd een toename van obesitas, hetgeen mogelijk een andere factor is die het ontstaan van zenuwschade bij mensen met pernicieuze anemie kan versnellen.
Behandeling
Ook over de behandeling is professor Biemond zeer duidelijk: “Zodra ontdekt was, dat leverpreparaten, liefst in injiceerbare vorm toegepast, een specifiek genezende invloed op de pernicieuze anaemie uitoefenden, werd ook de behandeling van de gecombineerde strengziekte met kracht ter hand genomen. Aanvankelijk waren de resultaten teleurstellend. Weliswaar kon de progressie van het neurologische beeld. worden gestuit en ook hier en daar vooral subjectieve verbetering worden bericht, doch er bleef een scherp contrast bestaan tussen het verrassende haematologische en het geringe neurologische resultaat. Beter werd de toestand, toen men voor het ruggemergslijden een belangrijk hogere leverdosis ging toepassen …… Sinds echter de laatste jaren het vitamine B12 is ontdekt, zijn de resultaten nog meer verbeterd, vooral wanneer de diagnose in een vroeg stadium wordt gesteld. Dan kan ongetwijfeld in vele gevallen een complete genezing worden bereikt. ….. Men passe dus thans dagelijkse intramusculaire injecties van B12 (15 gamma per ampul) toe, welke behandeling weken lang dient te worden voortgezet. …. Na afsluiten der behandeling is het noodzakelijk, dat bloedbeeld en voedingstoestand van het ruggemerg op peil worden gehouden door één injectie per week te blijven doorgeven. Dit moet steeds worden volgehouden, daar anders recidief mogelijk is. Maakt de patiënt een intercurrente ziekte of een periode van extra vermoeienis door, dan moet het aantal injecties tijdelijk worden verhoogd.”
Tot zover enkele citaten uit de lezing van professor Biemond.
Switch
Veel mensen hebben de afgelopen jaren ervaren dat hun neurologische klachten geweldig toenamen nadat zij -min of meer gedwongen- moesten overstappen van B12 injecties naar tabletten. Ten tijde van professor Biemond waren orale B12 preparaten niet beschikbaar, anders dan in de vorm van rauwe lever. Nu koop je een potje B12 tabletjes bij een drogist, maar er is geen enkel bewijs dat bij mensen met neurologische klachten, orale B12 suppletie even goed helpt als intramusculaire injecties. De observaties van Biemond waren heel duidelijk, lees maar wat hij schrijft over de behandeling: voor adequate behandeling van mensen met neurologische klachten zijn veel hogere doseringen van B12 nodig dan voor de behandeling van de bloedarmoede! En dit komt bijvoorbeeld terug in de bijsluiter van goed gedocumenteerde B12-preparaten. De volgende afbeelding is een deel van de tekst van de bijsluiter van Erycytol, een injecteerbaar vitamine B12 preparaat dat op de markt is in o.a. Oostenrijk.


Het is momenteel breed geaccepteerd dat de behandeling van mensen met B12 malabsorptie per definitie bestaat uit behandeling met intramusculaire B12 injecties. Deze injecties worden in principe levenslang voortgezet. Deze ‘best practice’ is enkele jaren geleden door de Engelse organisatie NICE nog eens uitgebreid samengevat, en kunt u hier nalezen: https://www.nice.org.uk/guidance/ng239
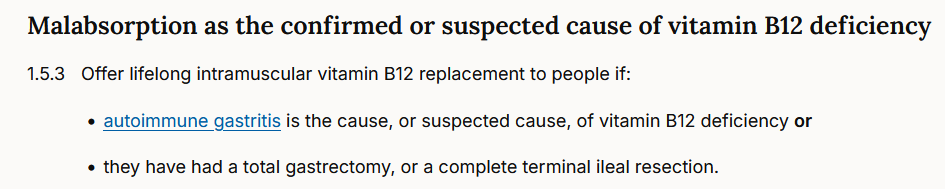
De frequentie van de B12 injecties op de lange termijn wordt dan bepaald op basis van anamnese, en staat onder regie van de persoon met B12 tekort zelf: klachten dienen te verdwijnen en weg te blijven. In de regel komen mensen uit op een frequentie variërend van 1 a 2x per week tot 1x per 4 a 5 weken. De frequentie van B12 injecties wordt NIET gebaseerd op bloedbepalingen. Hierbij dient te worden aangetekend dat bij mensen met een ernstige deficiëntie of een langdurig onbehandelde deficiëntie vaak (vooral cognitieve) restklachten blijven bestaan. Een survey onder meer dan 1000 leden van de Engelse Pernicious Anaemia Society leerde dat 30% van hen éénmaal per week of vaker B12 injecteert om de klachten te bestrijden.
Engelse experts beschrijven dit als in een recent artikel als volgt:
The guidelines’ emphasis on a symptom-based approach to treatment is an urgent and necessary shift that could positively influence current practice. They also stress the importance of individualised care, suggesting that B12 replacement dosage, frequency, and delivery method may need to be tailored to ensure treatment efficacy. However, many patients require treatment more frequently than current guidelines suggest. Survey data indicates that up to 50% of patients require more frequent injections to manage their symptoms effectively (Thain et al., 2024, DOI: 10.1101/2024.08.30.24312837). This is consistent with findings from Hooper et al., where 65% of patients received B12 injections according to current guidelines (every 2–3 months) [4]. Further qualitative evidence from interviews also highlights that many PA patients considered the current guidelines too restrictive, with symptoms reoccurring well before the next planned injection [24]. Bron: https://pubmed.ncbi.nlm.nih.gov/39984701/
door bhrw | jan 16, 2026 | B12, UK Biobank, wetenschap |

Scientists have identified the first DNA variant associated with a higher risk of developing B12 deficiency when a person is treated with metformin.
The analysis, which was done by the authors, identified a genome-wide non-synonymous SNP in the cubilin gene (CUBN, rs1801222/p.S253F), which was significantly associated with metformin-induced vitamin B12 deficiency. This finding was replicated in three Scottish cohorts, in the Diabetes Prevention Program Outcomes Study (DPPOS) cohort, and in a small clinical cohort from Liverpool. Vitamin B12 deficiency occurred in 0.84–1.20% of individuals who were not exposed to metformin regardless of their rs1801222 genotype. However, a large interaction with metformin use was observed, with vitamin B12 deficiency developing at 6.02% in GG, 7.96% in GA and 12.84% in AA genotype groups.
Co-author Ewan Pearson: “The 14% of white population with AA genotype are 2.5 times more likely to require B12 replacement GG. Equates to 10% requiring B12 replacement by 11 years after starting metformin with AA genotype vs 21 years for GG. A large effect!”
It should be noted that the primary analysis was conducted among participants in the UK Biobank. Individuals were identified with a diagnosis of vitamin B12 deficiency and/or a record of B12 injection prescriptions, and metformin use was extensively documented. However, clinical diagnosis of B12 deficiency is often only done on the basis of abnormal B12 measurements, and -for instance- measurements of methylmalonic acid are rarely done. Furthermore, people who are using (multi)vitamin supplementation may not so easily be recognized as truly B12-deficient. So, the incidence of B12 deficiency may be higher than reported in the paper. Nevertheless, this is great work in the beautiful dataset of UK Biobank, and confirmed in other cohorts, such as Generation Scotland, GoDARTS, and SHARE, and DPPOS. Researchers are eagerly awaiting the time when UK Biobank data will also be enriched with serum B12, MMA, and homocysteine measurements for all participants.
Full source: https://link.springer.com/article/10.1007/s00125-025-06655-5
For translation into Dutch, copy this blog to Google Translate
door bhrw | okt 17, 2025 | diabetes, wetenschap
The title of this blog contains two abbreviations, but important ones. IAH is impaired awareness of hypoglycaemia, and AGRP (or AgRP) is agouti-related protein. They are central in a paper just published by my talented colleague, Dr. Rita Varkevisser.

Learn how she summarizes the importance of this paper:
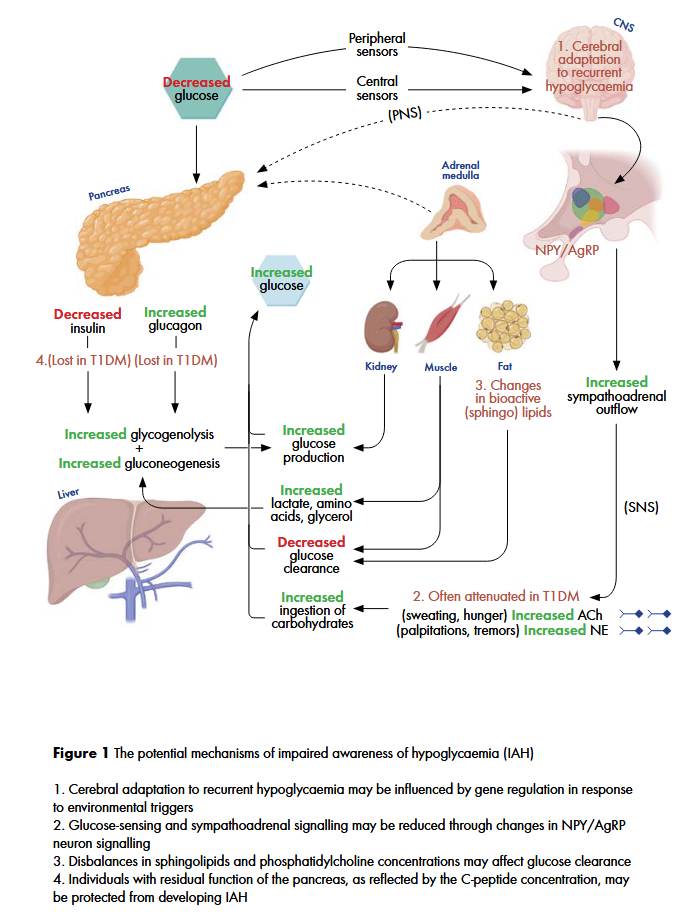
A compelling finding of this study was the association between agouti-related protein (AgRP) and impaired awareness of hypoglycaemia. AgRP is an important neuropeptide in energy homeostasis and is expressed in the adrenal gland and hypothalamus, specifically in the arcuate nucleus (ARC) and ventral hypothalamic nucleus (VMH) where central glucose sensing occurs. These glucose-sensing neurons in the ARC can influence glucose homeostasis through the activation of the autonomic nervous system and exert orexigenic effects through release of AgRP which inhibits MC4R neurons that stimulates food intake. The lower circulating plasma AgRP in individuals with IAH could suggest that these individuals could be less responsive to lower glucose levels. This may be due to threshold changes in the glucose-sensing neurons in the ARC and less hypothalamic-pituitary-adrenal (HPA) activation leading to less AgRP release from neuroendocrine cells in the adrenal medulla.
Another mechanism that may reduce AgRP release is the inhibition by other hormones such as insulin and leptin. Higher circulating levels of insulin usually leads to a reduction in appetite. In some individuals, hypothalamic insulin resistance may lead to increased food consumption despite higher circulating insulin levels. As a consequence of this insulin resistance, these individuals may be protected from hypoglycaemia, as the appetite suppression that would normally occur is no longer present. Although it is not yet clear how recurrent hypoglycaemia can influence glucose sensing and ARC/VMH mediated counterregulatory responses, further research into AgRP may help to elucidate the pathophysiology of IAH.
The mechanisms which dr. Varkevisser summarizers to play a role in IAH, are shown in this figure:
Dr. Varkevisser successfully defended her scientific thesis in November 2024. The full thesis with all her articles can be found on the University of Groningen website: https://research.rug.nl/en/publications/a-balancing-act-navigating-cardiovascular-vigilance-and-hypoglyca













 So, if you do not have anaemia, you don’t have a B12 deficiency, and outside the setting of ‘anaemia’, B12 is useless. This is absolutely not correct. Vitamin B12 is a very important compound, it has multiple effects in our body, and is especially needed for optimal nerve function and for well-functioning mitochondria, the small organelles in our cells that are responsible for making energy.
So, if you do not have anaemia, you don’t have a B12 deficiency, and outside the setting of ‘anaemia’, B12 is useless. This is absolutely not correct. Vitamin B12 is a very important compound, it has multiple effects in our body, and is especially needed for optimal nerve function and for well-functioning mitochondria, the small organelles in our cells that are responsible for making energy.















Recente reacties